OSTAR Proudly Introduces Their OSTAR P2 via an Indiegogo Crowdfunding Campaign

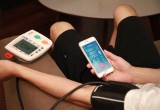
Toronto, Canada, June 1, 2016 (Newswire.com) - OSTAR Meditech, a Taiwan based company with over a decade of experience in manufacturing high quality medical devices, has launched a crowdfunding campaign on Indiegogo. This campaign has a funding goal of $60,000, which needs to be raised in order to bring the company’s newest innovation, the OSTAR P2, to market. The OSTAR P2 is a fully patented and FDA, CFDA and TFDA approved heart spectrum blood pressure monitor intended for home use. The idea behind this device is to help people keep a better eye on their health before they experience a life-threatening emergency. The team states, “We believe shortening the distance between patients and doctors is the keystone to truly achieve early warning for fatal diseases.”
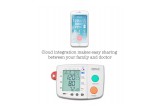
The OSTAR P2 has been equipped with features that are based on the team’s extensive medical experience. They tried to think of every need a user might have, and created their device to fit those needs. One of the most beneficial advances they have implemented on this monitor is improved reliability and accuracy. Standard blood pressure monitors often give incorrect readings because environmental factors can effect the accuracy. Most hospitals have technical teams just to constantly calibrate their devices to make certain their devices are accurate. OSTAR knew there was a better way, and equipped their devices with patented Auto-Calibration technology that not only checks, but calibrates the device every time a user turns it on. This feature makes the OSTAR P2 more effective and accurate than competitors on the market. The device also features a cloud monitoring system which increases the sharing and accessibility ease of the health data, and works in conjunction with a user friendly app that is currently under development.
"At OSTAR we have spent years with our trusted partners developing our medical devices, includes hospitals and universities. We implemented features with over a decade of medical device innovation experience."
OSTAR, Team
The team at OSTAR Meditech has been working very diligently on all of the essential groundwork for their OSTAR P2, and they are now ready to begin the process of bringing the device to market which is why they have launched their Indiegogo campaign, and hired a team of crowdfunding specialists to help them spread the word. As an added bonus with this campaign, the team is offering several perks. These perks, which serve as funding incentives, range in value from a “Keep in Touch” perk that enables a supporter to follow the campaign and receive all of the updates, to a “Thank You Package” and the opportunity to pre-order the OSTAR P2 at a substantial savings off of regular retail pricing. They even have an “exclusive to early adaptors” offer which gives campaign supporters the opportunity to complete their set by adding the OSTAR Oxymeter, a device that measures oxygen saturation making it the perfect companion to the P2. The team has a projected perk delivery date of November 2016. Full details can be found on their Indiegogo campaign page.
About OSTAR Meditech Inc.:
OSTAR Meditech was founded by Dr. Chang and is based in Taiwan. The company has over 14 years of experience in medical device innovation, development and manufacturing. Their hospital-grade products are currently used in hundreds of hospitals and long care centers in Taiwan and the U.S. Last year Dr. Chang decided he wanted to bring his technology to the consumer market, which led to creation of the OSTAR P2.
To learn more about the OSTAR P2 crowdfunding project Click Here
For more information about OSTAR Meditech Click Here
To contact via email write to Andy Peng at: andypeng@ostarhealth.com
For more information about the specialists at Smart Crowdfunding Click Here
Source: OSTAR Meditech Inc.