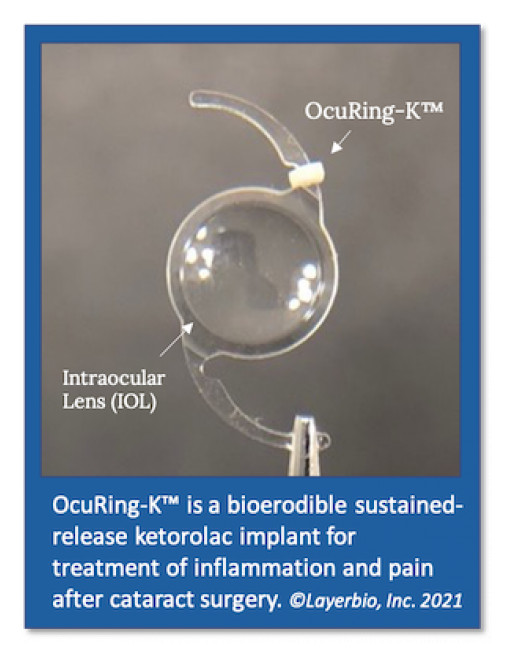
Category Ophthalmology and Optometry

Investigational Optejet® Microdose Dispenser from Eyenovia Shows Promise in Improving Treatment for Myopia, Presbyopia and Patients Getting Eye Exams
The Optejet® ophthalmic drug device is designed to precisely administer medication to the eye, potentially treating patients' conditions through gentler, more efficient dosing.
Read Press Release
NovaSight Announces FDA 510(K) Clearance of CureSight™ Digital Amblyopia Therapy
FDA 510 (K) clearance was based on a pivotal clinical trial evaluating the safety and effectiveness of this novel eye-tracking-based amblyopia therapy vs eye patching, the gold standard.
Read Press Release
Eyenovia's Novel Optejet® Dispenser Puts People's Eyes First
While traditional eye drop bottles require hand-eye coordination and deliver four to five times more medication than the human eye can hold, the Optejet® dispenser's human-centric design administers an easier, more accurate and appropriate dose directly where it is needed in the eye.
Read Press Release
Evo Visian® ICL Launches in the U.S.; an Evolutionary Vision Correction Procedure to Treat Nearsightedness is Now Available at Coastal Vision Medical Group
Leading Ophthalmology Practice, Coastal Vision Medical Group Among First to Offer EVO Visian ICL in Southern California to Provide Visual Freedom from Glasses and Contact Lenses
Read Press Release
Olympic Ophthalmics Announces FDA Clearance and Availability of Its Next-Generation iTEAR100 Neurostimulation Device, Expanded Patent Portfolio, and Further Clinical Studies
The newly approved connected device will facilitate patient access and communication between patients and eye care providers, collect data, and provide a platform between home and office to optimize treatment for dry eye
Read Press Release
Glauconix Data Published in Prestigious Ophthalmology Journal as Part of Their Work With NCX 667, a Novel Nitric Oxide Donor Agent With Intraocular Pressure Lowering Ability
The in-vitro studies performed in Glauconix's co-culture 3D conventional human outflow model confirm efficacy
Read Press ReleaseGlauconix Biosciences Awarded National Science Foundation Phase IIB Small Business Innovation Research Grant
3D Human Tissue Retina Vasculature model provides a new and novel approach to rapidly and efficiently screening numerous therapies for diabetic retinopathy and diabetic macular edema.
Read Press Release
NovaSight Announces First-Ever Pivotal Study for Lazy-Eye Digital Treatment Against Gold Standard Patching
NovaSight has announced that it is running a large, pivotal multi-center randomized controlled trial to assess the safety and effectiveness of an eye-tracking-based treatment for Amblyopia (Lazy Eye).
Read Press Release
Glauconix Biosciences Expands Scientific Advisory Board for 3D Human Tissue Retina Vasculature Model With the Addition of Leading Expert Dr. Patricia D'Amore
Appointment reflects Glauconix Biosciences' expansion of their 3D Human Tissue model platform within the Ophthalmology space and brings their second commercial model closer to market
Read Press Release