Category Diagnostics
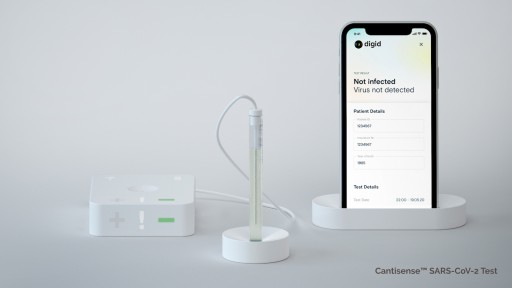
First Rapid Corona Antigen Test Approved for Clinical Study in Germany
The health tech company Digital Diagnostics AG from Mainz, Germany, has received approval for clinical trials from a German ethics committee and the Federal Institute for Drugs and Medical Devices (BfArM). This is a crucial regulatory step in the ongoing approval process for the Digid Cantisense™ SARS-CoV-2 test in Germany. The aim of the clinical trials is to demonstrate the reliability of the new technology in comparison with conventional PCR tests. The new test is particularly suitable for access control at airports or at large-scale event
Read Press Release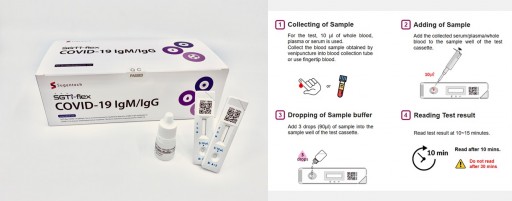
South Korean IVD Company, SUGENTECH's, COVID-19 IgM-IgG Rapid Test Listed on FDA
SUGENTECH's COVID-19 IgM&IgG 5-10 minutes rapid test kit is listed on the U.S. FDA's database and can be used in the U.S., as stated in Section IV.D of the FDA's Policy for Diagnostic Tests for Coronavirus Disease-2019, and is planning to go further to seek EUA from U.S. FDA.
Read Press Release
Flow Cytometry-Based Tools to Enrich Your Preclinical Immuno-Oncology Research, New Webinar Hosted by Xtalks
Flow cytometry provides a highly adaptable and quantitative method for investigating both the cellular distribution and intracellular changes that occur in tumors and peripheral lymphoid organs in response to therapy.
Read Press Release
FDA Perspectives on Computer Simulations in the Evaluation of Medical Devices, New Webinar Hosted by Xtalks
Live discussion with FDA and Siemens to provide an overview of the use of individualized computer simulation to predict clinical outcomes in medical device trials
Read Press Release