Category Pharmaceuticals and Biotech

Brainify.AI Joins Johnson & Johnson Innovation JLABS
Brainify.AI, an innovative company leveraging artificial intelligence to revolutionize the treatment of depression, is excited to announce it has joined Johnson & Johnson Innovation JLABS (JLABS) as a resident company. This marks a significant milestone for Brainify.AI and is another indication of the value and promise the company brings to the medical AI field.
Read Press ReleaseHealing the Wounds of Inflammatory Bowel Disease
A University of Chicago team identified a special type of stem cell that heals damaged bowel tissue in mice, a discovery that may have direct application to humans with inflammatory bowel disease. These early findings, made possible with seed funding from the GI Research Foundation, resulted in a recent five-year, $2.5 million grant from the National Institutes of Health to study those cells in humans.
Read Press ReleaseIschemix, Inc. Completes Successful Phase 1 Trial of Novel Compound to Treat Traumatic Brain Injury (TBI); Preparing for Conduct of Phase 2 Trial
CMX-2043, a novel, proprietary cytoprotective drug candidate, demonstrated excellent safety and tolerability in a Phase 1 single ascending and multiple ascending dose (SAD-MAD) clinical trial. CMX-2043 has previously demonstrated safety and efficacy in extensive preclinical pharmacology, toxicology and TBI translational studies. CMX-2043 will be Phase 2-ready by the first quarter of 2024.
Read Press Release
assisTek Announces Updated CORE Platform Release for Fall 2023
PA-based clinical technology provider assisTek has announced new features to its upcoming software release, including AI Integration, a Proprietary OS, SSO & Remote Provisioning, to add to its growing suite of eCOA & ePRO solutions.
Read Press Release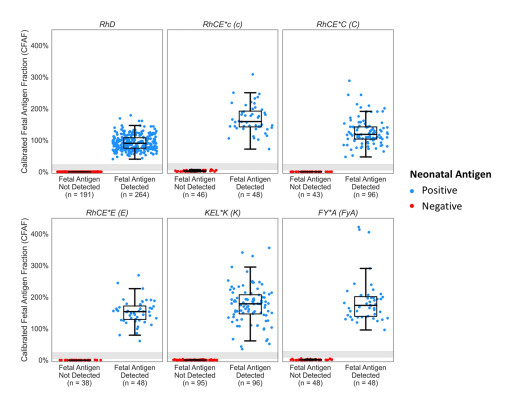
BillionToOne Publishes UNITY Fetal RhD and Fetal Antigen NIPT Clinical Validity Demonstrating >99.9% Accuracy
In a clinical validation study published on Scientific Reports, UNITY RhD NIPT and Fetal antigen NIPT demonstrated 100% sensitivity, 100% specificity, 99.9% precision with 0.1% no-call rate across 3,921 NIPT assays.
Read Press ReleaseiTolerance, Inc. and Northway Biotech Announce Partnership Agreement for Manufacturing of Streptavidin-FasL Fusion Protein for Innovative iTOL-100 Immunomodulatory Technology
iTOL-100 technology has broad applicability and can be applied to both allogenic and stem cell-derived organoids across a number of diseases.
Manufacturing agreement supports advancement of iTolerance's development of iTOL-100 towards first in human clinical study for lead program, iTOL-102.
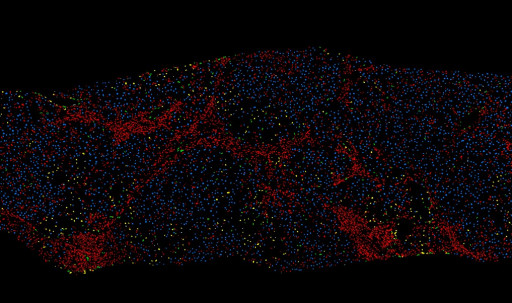
PharmaNest Launches Its Digital Pathology Spatially Resolved, Quantitative Tissue Panels to Quantify Tissue Injury and Inflammation in the Context of Fibrotic Conditions
PharmaNest, a leader in high resolution quantitative Digital Pathology and Artificial Intelligence, announces today the launch new quantitative tissue panels to properly quantify tissue injury and inflammation in the context of multiple fibrotic conditions, from discovery models to clinical development. Such spatially resolved datasets can be fused with other spatially resolved biology methods.
Read Press Release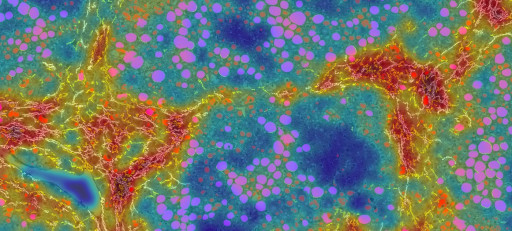
PharmaNest to Present Six Digital Pathology Abstracts at the International Liver Congress-EASL 2023, Including Liver Related Clinical Outcomes Prediction Results in NASH
PharmaNest, a leader in high resolution, single-fiber, quantitative Digital Pathology and Artificial Intelligence, showcases the utility and performance of its FibroNest Ph-FCS continuous fibrosis biomarker for the quantification of the severity of fibrosis and prognostics of liver related clinical events in NASH.
Read Press Release