Zylast Outperforms Alcohol Based Sanitizer in Hospital Controlled Clinical Trial
LAKE FOREST, Calif., October 4, 2017 (Newswire.com) - A 39-month study demonstrated the superior performance of a water-based hand sanitizer (Zylast® Lotion Antiseptic), which provides immediate and persistent effects, as compared to an alcohol-based hand sanitizer (ABHS) in a 347-bed hospital. This study, “Reduction of hospital-acquired infections with the use of a hand sanitizer with both immediate and persistent antimicrobial effect” was published in the Open Journal of Infectious Diseases and Epidemiology. Last month, Innovative BioDefense announced its first U.S. patent on this technology and product.
The hospital regularly collects data on Hospital Acquired Infections (HAIs) defined by the Center for Disease Control as an infection occurring at least 48 hours after admission. No changes were made in the data collection procedures. The alcohol dispensers were replaced with lotion dispensers throughout the hospital. The Zylast product in previous studies had been shown to kill bacteria in 15 seconds and continue to kill more than 99% of germs tested at six hours.
While using the ABHS, the hospital reported 10.3 HAIs per month dropping to 8.1 HAIs per month with lotion sanitizer, a statistically significant reduction of 20%. The most common HAIs were reported as C. difficile, which averaged 4.6 per month in the control group and reduced to 3.3 per month in the experimental group, a statistically significant drop of 28 percent. Tests have not been conducted to determine if Zylast can kill C. difficile in the spore state. There was also a significantly significant reduction in Vancomycin-resistant Enterococcus (VRE).
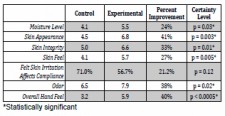
In addition, the staff was also given a survey comparing moisture level, skin appearance, skin integrity, skin feel, compliance, odor and overall hand feel. Using ABHS, 71 percent of healthcare workers reported that skin irritation made compliance with hand protocols more difficult compared with 57 percent with lotion. Overall hand feel showed a 40 percent improvement and was highly statistically significant.
Innovative BioDefense currently markets a Zylast Surgical Scrub, Antiseptic, Antiseptic Lotion and Foaming Soap utilizing the technology described in the patent in sizes ranging from 0.5 oz. to 1000 ml in automatic and manual dispensers.
For more information, please visit www.zylast.com. Zylast is a Trademark of Innovative BioDefense.
Contact: Innovative BioDefense, Inc. info@zylast.com 888-306-0316
Source: Innovative BioDefense