ZetrOZ Systems' sam® Ultrasound Technology Expands Clinical Education on Sustained Acoustic Medicine
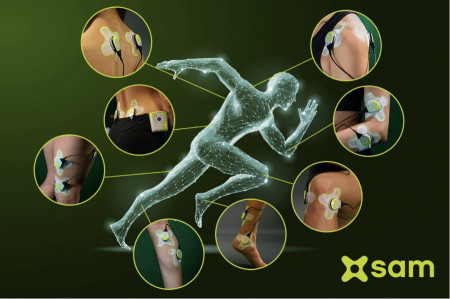
TRUMBULL, Conn., October 10, 2022 (Newswire.com) - ZetrOZ Systems, creator and manufacturer of the sam® wearable ultrasound device product line, is continuing to expand its clinical education program on the benefits of sustained acoustic medicine for soft tissue healing, the usage of the pain relief treatment and attaining health insurance coverage of the pain relief device.
Sustained Acoustic Medicine (sam®) is the only home-use bioregenerative medical device cleared by the U.S. Food & Drug Administration for the treatment of select medical indications, most commonly for chronic joint pain of the shoulder, knee and elbow, and the treatment of overuse injuries. More than 50 clinical and scientific studies support its effectiveness in healing injuries, restoring function and returning patients to work and everyday activities.
"Our sam® technology is touching thousands of lives daily, and we want to continue providing education to more healthcare providers as a way to bring this innovative intervention forward to Americans who can benefit from this treatment," said Dr. George Lewis, founder and CEO of ZetrOZ Systems. "This technology offers results without the need for opioids or other drugs while treating and healing patients up to 40% faster than standard methodologies."
Clinical education on sam® is directed by Dr. Rod Walters and Dr. Rajiv Mallipudi. Walters is a nationally recognized leader in athletic training with decades of experience at major universities and as a consultant to clients including the National Football League, the Big 12 Athletic Conference and Cirque du Soleil. He was inducted into the National Athletic Trainers Association Hall of Fame in 2005.
Dr. Mallipudi, MD, M.H.S, is a board-certified internal medicine hospitalist physician and assistant clinical professor at the Yale School of Medicine in New Haven, Conn. He is an active clinical researcher on healthcare quality improvement, with projects examining methods to reduce length of stay for hospital patients and to improve patient outcomes.
Walters and Mallipudi have a shared goal of educating physicians, physical therapists and athletic trainers about the benefits and use of the sam® device, in hopes of reducing the need for surgeries and pain medication and improving patients' likelihood of returning to work, sports and normal daily activities. By providing clinical training resources, including online videos, medical professionals now have information on best practices for the drug-free and non-invasive treatment of patients' injuries.
Adherence to a sam® regimen across the repair phase provides a necessary and safe intervention to ensure patients avoid chronic, recurring injuries. Recommended practices are specified in the education materials and include:
- The sam® treatment can be applied with one or two ultrasound stimulators.
- The sam® device should be applied a minimum of four times a week.
- Treatments should be applied incrementally to assess patients' response to treatment, from an initial one-hour treatment to a maximum of four hours per treatment.
- For acute conditions, treatment duration can range from 4 to 8 weeks.
- For chronic conditions, treatment can range from 2 to 12 weeks.
Clinical education instructs physicians that sam® can serve as a stand-alone treatment or be used to supplement other treatment approaches to stimulate soft-tissue repair and that its value is as a treatment option that does not require surgery or pain relief medications.
The sam® treatment also is a cost-effective treatment option. For example, a 2020 study on knee osteoarthritis, published in the Journal of Orthopaedic Surgery and Research, found sam® provided a greater improvement in functional effectiveness per dollar of care than the standard care options.
ZetrOZ Systems continues to provide education on sam® technology, most recently participating in the annual PHATS (Professional Hockey Athletic Trainers Society) SPHEM (Society of Professional Hockey Equipment Managers) 2022 Conference in June. Findings showed sam® is a non-invasive, non-narcotic alternative to pain, and that the device's ease of use led athletes to return to normal activity after treatment.
To learn more, visit zetrozsystems.com.
About ZetrOZ Systems
ZetrOZ Systems is leading healing innovation in sports medicine, developing wearable bioelectronic devices for the delivery of sustained acoustic medicine (sam®). Researched and funded by the federal government, ZetrOZ is built on proprietary medical technology of +46 patents and is the exclusive manufacturer and developer of sam®, a product line designed for the treatment of acute and chronic musculoskeletal conditions. To learn more, visit zetrozsystems.com.
Source: ZetrOZ Systems
