ZetrOZ Systems Expands Patient Access for Sustained Acoustic Medicine Treatment
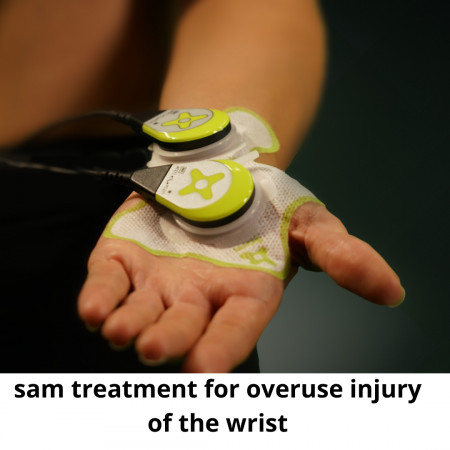
TRUMBULL, Conn., December 2, 2021 (Newswire.com) - ZetrOZ's sam® is the only FDA-cleared, long-duration, home-use, ultrasound treatment system, with over 30 peer-reviewed studies showing it effectively increases nutrient transport, relieves pain, restores function, and returns patients back to work, sports and other daily activities.
Its continuous ultrasound therapy promotes injury healing to deep tissue by inhibiting inflammation and increasing the rate of tissue regeneration, angiogenesis, and nutrient exchange. sam® was designed for easy, long-term home use. The sam® device is easy to use and is wearable, allowing patients to continue their daily routine.
The patient and health provider portal at samrecover.com details information for patients and physicians, physical therapists, chiropractors and other healthcare providers. Sections of the portal include clinical training, patient education videos, conditions treated and a large body of clinical evidence supporting the efficacy of sam® treatment for soft tissue injuries.
"Our sam® technology accelerates the healing process, reducing the need for potentially addictive opioids or invasive surgery," said Dr. George Lewis, founder and CEO of ZetrOZ Systems. "Our patient and health provider portal makes sam® easily accessible to everyone, including healthcare professionals who want to have more options for their patients dealing with soft tissue injuries and conditions like arthritis, back spasms, disk herniation, and tendonitis."
sam® is a Class II medical device and available by prescription.
To learn more, visit samrecover.com or zetrozsystems.com.
About ZetrOZ Systems
ZetrOZ Systems is an FDA cGMP and ISO 13585 medical technology company headquartered in the southern coastal region of Connecticut. The organization also has manufacturing facilities across the United States. ZetrOZ Systems produced UltrOZ®, sam®Sport and sam®Pro 2.0 to provide safe and effective treatment options for prevalent conditions such as arthritis. Learn more at zetroz.com and samrecover.com.
Media Contact:
Bianca D'Angelo
(203)577-7588 (Direct)
bianca@newswire.com
www.Newswire.com
Source: ZetrOZ Systems
