The Fighting for Kaiden Foundation and rareLife Solutions Announce New Partnership in Support of oneSMAvoice.com
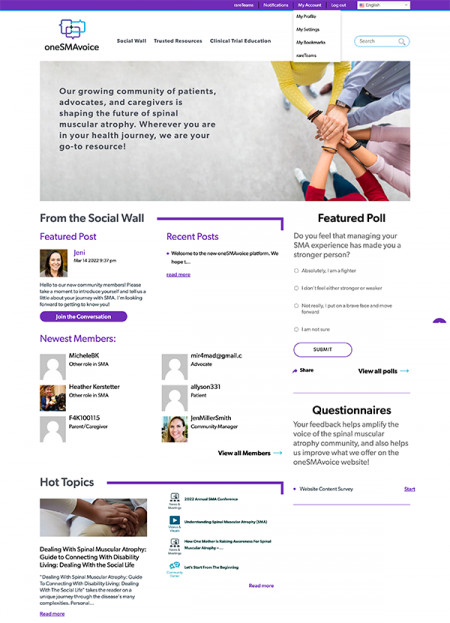
WESTPORT, Conn., August 1, 2022 (Newswire.com) - The Fighting for Kaiden Foundation and rareLife solutions have partnered to bring oneSMAvoice, a new online community and disease knowledge platform, to the spinal muscular atrophy (SMA) community.
The oneSMAvoice platform supports the needs of the SMA community by providing opportunities for social connection and access to trustworthy disease, treatment, and clinical trial information from across the web, vetted by SMA experts.
"This new relationship with rareLife solutions is an exciting step for our foundation," said Kristina DeFazio, co-founder, board chair, and executive director of the Fighting for Kaiden Foundation. "We are excited to introduce our members to the great resources that oneSMAvoice offers and, knowing how isolating it can feel to be a rare disease patient or caregiver, we look forward to seeing new relationships built through the community's social wall."
oneSMAvoice will benefit from ongoing collaboration among rareLife solutions, the Fighting for Kaiden Foundation, and advisors from the health care, advocacy, patient, and caregiver communities. Advisors to rareLife solutions' family of onevoice communities provide critical insight into the patient and caregiver experience and provide expertise that helps rareLife ensure that onevoice communities offer only the best and most trustworthy resources.
"We are thrilled to be partnering with the Fighting for Kaiden Foundation," said Dan Donovan, founder and chief executive officer of rareLife solutions. "They are a highly respected advocacy organization and we are excited to bring their knowledge, insight, and passion into our work on oneSMAvoice."
Among the tools and resources featured on oneSMAvoice are:
- Smart Social Wall, which provides a secure and welcoming space for community conversation while highlighting related curated resources from across the platform.
- Trusted Resources, a section devoted to providing vetted, trustworthy resources across popular topics, ranging from caregiver support to emerging treatments and beyond.
- My Clinical Trials, a locator tool that helps patients and caregivers find trials that meet their needs and connects them with clinical trial contacts to learn more.
- RareTeams, a private group messaging tool that allows for quick and easy conversation and knowledge-sharing with other members.
- Email newsletters, which bring the latest conversations on the Social Wall and featured resources into the members' email inboxes.
For treatment developers, onevoice community sponsorship provides a unique opportunity to share knowledge with rare disease communities, drive awareness of clinical trials, generate insights, and gather patient experience data (PED). Under the 21st Century Cures Act, the federal Food and Drug Administration (FDA) is creating a framework for the incorporation of PED into FDA filings. Now the FDA can consider other information, in addition to traditional clinical trial data, to help guide regulators in their assessments of new treatments.
For more information, contact Laura Wuerth at lwuerth@rarelifesolutions.com.
Source: rareLife solutions