Nooro Whole Body Massager Reviewed: Does It Really Work for Neuropathy, Foot Pain, and Poor Circulation?
New analysis explores FDA-cleared NMES device category, research-backed circulation mechanisms, and key considerations for individuals evaluating non-pharmaceutical foot wellness tools
DOVER, Del., April 23, 2026 (Newswire.com) - Disclaimers: Neuropathy, nerve pain, and circulation-related conditions should be evaluated by a qualified healthcare professional. Always consult a licensed healthcare provider before beginning any new device-based regimen, particularly if you have existing health conditions, take medications, or have an implanted electronic medical device. This article contains affiliate links. If you purchase through these links, a commission may be earned at no additional cost to you. This compensation does not influence the accuracy or integrity of the information presented. For the most current product details, pricing, and policies, verify directly at the official Nooro website.
Nooro Whole Body Massager Review 2026: What Consumers Should Know About NMES Technology, Circulation Support, and At-Home Use
You saw the ad. Maybe it was Facebook, maybe YouTube, maybe Instagram. Someone pressing a small device against the bottom of their foot and describing what it was like to finally wake up without that familiar burning and tingling in their feet.
And now you are here, running a quick search to find out whether the Nooro Whole Body Massager actually delivers, or whether you are looking at another polished ad campaign wrapped around a product that goes nowhere.
That instinct to verify before you spend is the right one. This guide was built for exactly that moment.
What follows is a detailed review based on publicly available information: the brand's official product page, their legal pages and terms, a peer-reviewed study they cite, live SERP research on competing articles and claims, and the brand's own FDA documentation. We address every major question people search when they are deciding whether this device makes sense for their situation.
If you have diabetic neuropathy, peripheral neuropathy, burning or tingling feet, poor lower-leg circulation, or swollen ankles, this guide was written for you. If you are buying this as a gift for a parent with foot pain, there is a section for that too. And if you have already tried TENS units, medications, or physical therapy and want to know whether NMES is genuinely different, we cover that directly with the research behind it.
Check out the Nooro Whole Body Massager on the official website
Disclosure: If you buy through this link, a commission may be earned at no extra cost to you.
Let us start with the question almost everyone is asking when they arrive here.
Is the Nooro Whole Body Massager Legit?
This is the first thing most people want answered after seeing the ad, so here it is before anything else.
Based on a full review of the brand's official materials, legal documentation, FDA records, and publicly available consumer feedback: the Nooro brand is a legitimate direct-to-consumer operation built around a device that holds confirmed FDA 510(k) clearance as a Class II medical device. The underlying NMES technology it uses has genuine peer-reviewed research supporting the circulatory mechanism it operates on. The brand publishes verifiable corporate information, a phone number, email support, and a 90-day money-back policy documented in official terms.
That is what the evidence supports.
What honest research also turns up: the brand's parent company, XF Agencija US Inc., carries an F rating with the Better Business Bureau, with publicly visible complaints around product effectiveness and refund processing. This does not make the technology fraudulent, but it is information any buyer should factor into their decision, and it is exactly why this guide covers the guarantee terms and return process in detail rather than letting the marketing language do all the work.
The device is real. The technology has FDA clearance. The outcomes for any individual are variable. The customer service history has documented friction. That is the complete honest picture, and the rest of this guide operates from that foundation.
This article is educational and does not constitute medical advice. Consult your physician before using any electrical stimulation device.
What Is the Nooro Whole Body Massager?
The Nooro Whole Body Massager is a direct-to-consumer neuromuscular electrical stimulation (NMES) device sold by XF Agencija US Inc. under the Nooro brand. According to the brand's official product documentation, the underlying device is identified as "Sumsens TENS and PMS," model SM9187, manufactured by Hang Zhou Anqing Shen Zhen Electronics Limited and branded under the Nooro name for the US market.
The device is designed for at-home adult use and delivers low-level electrical impulses through adhesive sticky pads placed on the feet, ankles, and calves. According to the brand, the intended mechanism centers on stimulating blood circulation in the lower extremities by activating the calf musculature.
Here is how that mechanism works in plain terms: the NMES electrical signal causes the calf muscles to contract. Those contractions activate what the research literature calls the musculo-venous pump, the same mechanism that occurs naturally when you walk. That pump moves blood up from the lower legs toward the heart while drawing oxygenated blood back down to the peripheral tissues and nerves of the feet. The brand's premise, grounded in the circulation research they cite, is that supporting this process may contribute to perceived comfort for people experiencing neuropathy-related foot discomfort. This is the circulation and general wellness mechanism the device is designed to support. It is not a treatment claim.
The Nooro Whole Body Massager is marketed as an FDA-cleared Class II medical device. According to publicly available FDA records and the brand's own product documentation, the device received 510(k) clearance under submission K192248 in 2019. FDA 510(k) clearance means the agency determined the device is substantially equivalent to a legally marketed predicate device in terms of safety and effectiveness for its cleared indication. It does not validate specific marketing claims made about the device by the brand, and it does not mean the FDA independently tested this specific product for neuropathy relief. Consumers who want to verify the clearance independently can search the FDA's 510(k) database using the submission number K192248.
Consult your physician before use, particularly if you have cardiovascular conditions, implanted electronic devices, or are pregnant.
Read: Nooro Whole Body Massager Claims Evaluated
NMES vs. TENS: Why the Technology Difference Matters
One of the most common questions from people who have already tried TENS units is whether NMES is genuinely different or just the same thing with a new name. The answer matters for how you evaluate this device.
TENS stands for transcutaneous electrical nerve stimulation. NMES stands for neuromuscular electrical stimulation. Both use low-level electrical current delivered through surface electrodes. The difference lies in what kind of nerve fiber each targets and what physiological response it produces.
TENS primarily targets sensory nerve fibers. The most commonly cited mechanism is gate control theory, where stimulating sensory nerves at certain frequencies interferes with pain signal transmission to the brain. TENS can provide temporary relief during and after use, but it does not typically produce meaningful muscle contraction and does not engage the circulatory pump mechanism in the same way.
NMES targets motor nerve fibers. The signal parameters are set to produce actual involuntary muscle contractions. In the foot and calf context, this means the calf muscle physically contracts during the session, activating the musculo-venous pump. That circulatory response is the mechanism NMES is built on.
NMES and TENS operate on meaningfully different pathways. Whether one is more appropriate for a particular person depends entirely on their specific situation and is a question for their physician. The practical point is that they are not the same technology. If you have used a TENS unit without lasting benefit, the mechanistic difference between NMES and TENS is a scientifically grounded reason to evaluate NMES separately, rather than assuming the outcome will be the same.
The brand's comparison chart positions the Nooro device as offering muscle strengthening, spasm prevention, range of motion support, and muscle atrophy prevention that standard TENS units do not. This comparison is the brand's own marketing positioning and not an independent clinical head-to-head result. The underlying technological distinction between NMES and TENS, however, is supported in the rehabilitation literature.
The Research Behind NMES Foot Stimulation
Before evaluating the Nooro device specifically, here is what peer-reviewed research says about the technology category.
The brand cites a specific study on their product page: "The effect of an 8-week treatment program using a novel foot neuromuscular electrical stimulator on physical function, leg pain, leg symptoms, and leg blood flow in community-dwelling older adults" by Binoy K., Darren T., and Tim W., published in the scientific literature and accessible via PubMed reference ID 36242050. According to the brand's summary of this research, the study found that NMES applied to the plantar surface of the feet was associated with changes in blood circulation in the legs during use, and was associated with reported changes in symptom measures including pain, heaviness, cramps, and tiredness, through activation of the musculo-venous pump.
This is a real peer-reviewed study. It is not invented.
The critical qualification this guide will maintain throughout: the cited study evaluated a specific NMES device in a specific study population over eight weeks under controlled research conditions. It does not constitute clinical validation of the Nooro Whole Body Massager specifically. These findings document what the NMES technology category has demonstrated under studied conditions. They do not prove that this specific finished consumer product will produce identical results for any individual user. This is technology-level research, not finished-product clinical trial data for the Nooro device.
What the broader NMES literature supports: NMES applied to the lower limbs consistently increases venous blood flow velocity during stimulation in study populations. The musculo-venous pump activation mechanism is well-established in clinical rehabilitation contexts, particularly in research on circulation support and deep vein thrombosis prevention. NMES devices are generally well-tolerated when used as directed, with primary contraindications centered on implanted electronic devices and active skin conditions.
The research foundation for the technology this device uses is real. Individual outcomes from this specific consumer product are variable and not guaranteed.
See current pricing on the official Nooro website
Who Is the Nooro Whole Body Massager Designed For?
The brand positions this device primarily for adults with peripheral neuropathy symptoms in the feet and lower legs. But the actual audience is several distinct groups, each with different reasons to evaluate this type of device. Here is an honest breakdown.
People with Diabetic Peripheral Neuropathy
Diabetic peripheral neuropathy is one of the most common forms of neuropathy and one of the most frequent reasons people land on this search. Elevated blood glucose over time can affect peripheral nerves, and the feet and lower legs are among the most commonly involved areas. Symptoms include burning, tingling, numbness, and in more advanced cases, reduced protective sensation.
Circulation to the peripheral extremities is also frequently affected in people with long-term diabetes, making the NMES circulatory mechanism particularly relevant to discuss with a care team in this population.
Important for this group: people with diabetes should consult their physician before using any electrical stimulation device. Diabetes affects skin integrity and sensation in ways that require specific precautions. A physician familiar with your history is the right person to assess whether NMES foot stimulation is appropriate for your case. This device does not replace diabetic foot care, glycemic management, or any prescribed medical treatment.
People with Peripheral Neuropathy from Other Causes
Peripheral neuropathy has many potential causes beyond diabetes: chemotherapy-induced neuropathy, autoimmune conditions, B12 deficiency, hereditary conditions, alcohol-related nerve changes, and in some cases, idiopathic neuropathy with no identified cause. The circulatory mechanism the brand focuses on is most relevant for neuropathy where vascular factors are involved. For neuropathy with other primary causes, NMES may offer general wellness benefits but does not address the underlying cause. Discuss with your physician.
People with Burning Feet, Tingling, or Numbness
Many people in this search category do not have a formal diagnosis. They have symptoms: feet that burn at night, persistent tingling, patches of numbness, or cold feet that never warm properly. These symptoms can reflect early neuropathic changes, circulation issues, or other conditions that warrant medical evaluation. If you have these symptoms and have not yet seen a physician, that evaluation should come before any consumer device purchase.
People Seeking a Drug-Free Daily Wellness Option
A significant portion of people searching for devices like this want a non-pharmaceutical addition to their management routine. A consumer NMES device operates on an entirely different pathway from medications like gabapentin or pregabalin, which work pharmacologically to modulate nerve signal transmission. These approaches are not interchangeable and should not be treated as substitutes for each other. Do not modify any prescribed medication based on using a consumer device.
Athletes and Active Adults
According to the brand's own FAQ, the device has been used for general post-activity recovery. This application is distinct from the neuropathy use case and reflects the calf muscle activation mechanism being relevant to exercise recovery, where circulation support to fatigued muscles matters. The brand's FAQ is the source for this application - it is not an endorsement by named athletes.
Gift Buyers
A meaningful portion of purchases come from adult children buying for a parent with foot pain, or spouses buying for a partner who suffers every morning. If that describes you: the device takes under three minutes to set up, requires no technical knowledge, and comes with a 90-day money-back guarantee on one-time purchases. For someone with chronic foot discomfort who has been reluctant to spend money on themselves, this is a practical gift in a technology category with genuine research roots. It is not a cure, but it is a thoughtful and credible tool.
For all populations: consult a physician before use, particularly if there are cardiovascular conditions, implanted devices, or pregnancy. This device does not replace professional medical management of any condition.
What Neuropathy Actually Does to Daily Life
Understanding this matters for calibrating expectations honestly and for recognizing whether this device is addressing the right problem for your situation.
For most people with significant neuropathic foot symptoms, the day starts with a negotiation. Getting out of bed means bracing for first contact with the floor. Mornings are often the worst window because the feet have been still all night, circulation has been minimal in the recumbent position, and the nerves have been firing in the absence of competing sensation. That first step onto a cold floor can feel like stepping on broken glass, electric current, or nothing at all - and all three of those sensations are disorienting in their own way.
Standing for any extended period becomes difficult to plan around. Activities that should be unremarkable - standing at a kitchen counter, walking through a grocery store, being on your feet at a family gathering - become events that require managing how the feet will feel before, during, and after. People begin calculating tradeoffs constantly, often without fully recognizing they are doing it.
The nights can be equally difficult. Neuropathic symptoms frequently intensify at night when there is less competing sensory input. Poor sleep compounds everything: fatigue lowers pain tolerance, reduces motivation for physical activity, and creates a cycle where the symptoms worsen the rest and the lack of rest worsens the symptoms.
The emotional layer of all of this is real and often underacknowledged in clinical conversations. Chronic foot discomfort compresses independence in ways that accumulate invisibly. People stop accepting invitations that involve standing or walking. They become reluctant to travel. They feel older than they are, limited in ways their peers are not yet experiencing. For many people managing diabetic neuropathy alongside a chronic condition, the foot symptoms become a central organizing fact of daily life.
This is the full context in which the Nooro Whole Body Massager is being evaluated. Not as a relaxation gadget. As a daily tool for people whose feet have become a genuine source of limitation, who are looking for something credible they can use at home without scheduling another appointment.
That context also sets the honest expectation. This device supports circulation as a general wellness mechanism. It does not eliminate neuropathy. It does not repair nerve damage. Some people who use it consistently report changes in how their feet feel during and after daily sessions. Others do not notice meaningful changes outside of device use. The ninety-day evaluation window exists precisely because the honest answer is individual and takes time to determine.
The Nooro Whole Body Massager as a Gift
If you arrived at this guide as someone buying for a parent, partner, or loved one with chronic foot pain, this section is for you.
When you give someone a foot device in a technology category with real research behind it and an FDA clearance on record, you are communicating something that a generic gift does not: that you have actually been paying attention. That you know the mornings are hard. That you looked into it before buying something.
Warmer weather has arrived and with it comes the specific frustration of wanting to be outside and active while feet make it complicated. A daily device that takes fifteen minutes, requires no appointments, and comes with a ninety-day trial window is a practical gift with genuine emotional resonance right now. It meets the person exactly where they are.
What to communicate when you give it: be honest that this is a device in a technology category with legitimate FDA clearance and peer-reviewed research backing - not a guaranteed cure. Let them know the ninety-day window is real for one-time purchases. Encourage them to mention it to their doctor at their next visit, so their physician is aware.
When purchasing as a gift, make sure to select the one-time purchase option rather than any subscription - only the one-time purchase is covered by the ninety-day money-back guarantee. The device ships from the US warehouse, takes under three minutes to set up, and requires no technical knowledge.
Get the Nooro Whole Body Massager as a gift on the official website
Nooro Whole Body Massager: Full Features and Specifications
According to the official Nooro product page and brand documentation as of April 2026:
Eight pre-programmed stimulation modes: The device comes with eight preset programs, described by the brand as pre-programmed with clinically studied parameters. Different modes produce different patterns and rhythms of stimulation.
Nineteen adjustable intensity levels: Users control stimulation strength across nineteen settings. The brand recommends beginning at lower intensity and building up gradually as comfort allows.
Adhesive sticky pads: The device attaches via reusable adhesive pads. According to the brand, the included pair is rated for approximately one month of daily use. Replacement sets are available at approximately ten dollars per pair, which the brand estimates at roughly forty-seven cents per session.
Wireless and rechargeable: The device charges via the included USB cable. The brand states the blue indicator light turns off when charging is complete.
Broad application range: While marketed primarily for foot and neuropathy-related use, the brand describes the device as a whole body massager with application areas including feet, ankles, calves, and other locations.
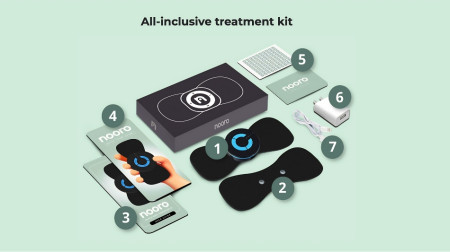
Kit contents per brand listing: The standard kit includes the device, two adhesive pads, a user guide with nine-step setup, a quick-start guide, a 90-day habit tracker with stickers, USB charger, USB cable, and a free eBook on foot pain remedies as a current promotional addition.
All specifications and kit contents are attributed to the brand's official materials as of April 2026 and are subject to change. Verify current inclusions at checkout.
How to Use the Device
According to the brand's official usage instructions, setup takes under three minutes.
Step one: Unbox and locate the host unit, USB cable, charger, sticky pads, user guide, quick-start guide, and habit tracker.
Step two: Connect the USB cable and charge fully. The blue indicator light turns off when complete.
Step three: Attach the host unit to the sticky pad, remove the protective film, press the power button twice to activate, and select a mode and intensity level.
Step four: Place the device on the target area for the session. The brand's primary recommendation is feet, ankles, and calves for the circulation-focused use case.
Daily protocol per the brand: Start with two five-minute sessions per day and build toward ten to fifteen continuous minutes. The brand recommends placing the device on feet, ankles, and calves together to engage the full lower-extremity circulation chain.
The brand's FAQ notes that some users do not notice changes in the first week and recommends continuing through week two before drawing conclusions. According to the brand, some individuals begin noticing changes after approximately two weeks of consistent daily use.
These timelines are the brand's usage pattern guidance. Individual experiences vary based on symptom severity, underlying condition, consistency of use, and individual health factors. The ninety-day window built into the money-back guarantee for one-time purchases exists because meaningful evaluation genuinely takes time.
Do not use on broken, irritated, or infected skin. Do not use while sleeping. Consult your physician before use if you have a pacemaker, ICD, or any implanted electronic medical device. Do not use if pregnant without physician clearance.
What to Realistically Expect Over 90 Days
The brand does not publish a guaranteed week-by-week progression. What follows draws on the brand's usage guidance and the general NMES research context, not clinical trial data for the Nooro device specifically.
First two weeks: Familiarization. Users become comfortable with modes and intensity levels, establish a daily routine, and experience the sensation of the device during sessions. The stimulation is perceptible during use. Some users report noticing changes in how their feet feel during or immediately after sessions within this window.
Weeks three through eight: This is the window the brand's FAQ and the cited eight-week PubMed study both align with as the relevant period for evaluating cumulative circulatory effects. Some users who maintain consistent daily use report beginning to notice changes in how their feet feel outside of device sessions during this stretch.
Beyond sixty days: Users who have maintained daily use are positioned to make a genuinely informed evaluation before the ninety-day guarantee window closes.
What this does not guarantee: changes within any specific timeframe, changes of any particular magnitude, or effectiveness for every presentation of neuropathic symptoms. Some users will find meaningful benefit. Some will not. The honest answer is individual.
These timelines draw on brand guidance and general NMES research context. They do not represent guaranteed clinical outcomes.
Nooro Pricing and What You Actually Pay
According to the official Nooro website, the brand's listed promotional price for the Nooro Whole Body Massager is $49.95. The brand lists a higher standard retail price alongside this promotional figure; as with most direct-to-consumer brands, promotional pricing should be verified at checkout, as these figures are subject to change without notice.
Free shipping is noted for qualifying orders. Replacement adhesive pads cost approximately ten dollars per set per month for daily users, per the brand's listing. The brand estimates approximately forty-seven cents per individual session.
Coupon codes: The brand does not publish standing coupon codes on the product page. Any third-party discount codes should be verified at checkout.
FSA and HSA eligibility: Coverage for consumer NMES devices under FSA and HSA plans varies by plan and administrator. The brand's materials do not make explicit FSA or HSA claims. Verify directly with your plan administrator.
Insurance: Many direct-to-consumer devices are not covered by traditional health insurance. Coverage varies by plan. Confirm with your insurer directly.
All pricing is attributed to the brand's official page as of April 2026 and is subject to change. Always verify current pricing at checkout before purchasing.
View current pricing and availability on the official Nooro website
The 90-Day Guarantee: What the Brand's Legal Pages Say
Because the guarantee terms and the marketing language differ in ways that matter, this section works directly from the brand's official returns policy and terms of sale rather than the sales page copy.
What is covered: The ninety-day money-back guarantee applies to one-time purchases of physical Nooro products bought directly through nooro-us.com. Contact support@nooro-us.com within ninety days of your order date to initiate the process. Refunds go to the original payment method.
What is not covered: Subscription products are explicitly excluded from the ninety-day guarantee. Monthly VIP Peak Health Club payments are noted as non-refundable. Free trials converting to paid subscriptions must be cancelled before the trial period ends. Cancel at least forty-eight hours before the next billing date to stop future charges.
Return shipping: Customers are responsible for return shipping costs. The brand does not provide prepaid return labels. At roughly ten to fifteen dollars in return shipping, the net financial risk of a trial is approximately thirty-five to forty dollars rather than zero - still a manageable evaluation window, but worth knowing before you order.
Processing time: The brand states processing takes seven to ten business days after receipt and inspection, with up to an additional thirty days for the refund to post depending on your bank.
Guarantee terms are based on the brand's official policies as of April 2026 and are subject to change. Always review current terms on the official Nooro website before purchasing.
Brand Transparency: What You Should Know Before Ordering
Operating entity
XF Agencija US Inc., registered in Delaware, address 1111B S Governors Ave Number 80212, Dover, Delaware 19904. EIN 93-1616450 is published in the brand's public legal documents. An international entity, XF Agencija Limited, is also listed in Hong Kong.
FDA clearance
According to the brand's official product documentation and publicly available FDA records, the Nooro NMES Foot Massager received 510(k) clearance as a Class II medical device under submission K192248 in 2019. The underlying device is identified as "Sumsens TENS and PMS," model SM9187, manufactured by Hang Zhou Anqing Shen Zhen Electronics Limited. This clearance applies to the device's safety profile within its cleared category - it does not validate specific marketing claims about neuropathy reversal or guaranteed pain outcomes.
BBB rating
XF Agencija US Inc. carries an F rating with the Better Business Bureau, with publicly available complaints centered on product performance and refund processing. This is factual information any buyer should know. It does not make the technology fraudulent, but it does reinforce why understanding the exact guarantee terms and return process before purchasing matters more than usual here.
Customer count
According to the brand's product page, Nooro reports serving over 70,000 customers. This is the brand's stated figure and has not been independently verified.
"Designed by a leading podiatrist" claim
The brand states the device was designed by a leading podiatrist. This is attributed to the brand's own marketing materials. MBK has not independently verified this credential. If it matters to your decision, contact the brand directly.
Third-party availability
The brand states the device is not sold on Amazon or eBay and warns that other companies misrepresent themselves as Nooro. Purchase only through the official website for authenticity and guarantee eligibility.
Regulatory disclaimer from the brand's own website
"The statements on this website have not been evaluated by the Food and Drug Administration. Nooro products are not intended to diagnose, treat, cure or prevent any disease."
Is It Safe? Who Should Not Use This Device
Do not use without explicit physician clearance if you have:
A cardiac pacemaker, implanted cardioverter-defibrillator, or any other implanted electronic medical device. Electrical stimulation can interfere with these devices. This is a firm contraindication across the NMES and TENS category.
Any form of heart disease. The brand's FAQ specifically recommends consulting a doctor before use if you have heart disease. Take this seriously.
Active deep vein thrombosis or thrombophlebitis. Stimulating circulation in a limb with active thrombosis requires physician evaluation first.
Epilepsy or seizure disorders. Electrical stimulation devices are generally contraindicated without medical guidance in this population.
Do not use on: broken, irritated, infected, or inflamed skin; areas with severely reduced or absent sensation without physician guidance (people with advanced neuropathic numbness may not be able to gauge whether intensity is safe).
Pregnancy: The brand recommends consulting a doctor before use. Electrical stimulation is generally not recommended during pregnancy without medical supervision.
For the general population without these contraindications: NMES devices at consumer intensity levels are generally well-tolerated. The sensation during use should be perceptible but not painful. If stimulation feels sharp or painful, reduce the intensity level. Some users report mild skin irritation from adhesive pads over time.
This safety overview is a general summary. Always consult your physician before use if you have any health condition or concern.
Nooro vs. The Alternatives: Honest Category Comparison
Nooro vs. prescription neuropathy medications
Medications like gabapentin, pregabalin, and duloxetine work through pharmacological modulation of nerve signal transmission - a completely different pathway from circulatory support. They require physician prescribing and carry their own tolerability considerations. A consumer NMES device and prescription neuropathy medication are not the same category of intervention and are not interchangeable. Do not discontinue or modify any prescribed medication based on using this device.
Nooro vs. professional physical therapy
Clinical NMES equipment in supervised physical therapy settings is used with individualized parameters, professional monitoring, and integration into a comprehensive treatment plan. The tradeoff with a consumer device is no clinical oversight and no adaptive protocol - but also no scheduling, no copays, and daily availability at home. For people who need supervised professional care, PT is the appropriate primary path. For those already in a stable routine looking for a daily at-home supplement, a consumer device can complement that care.
Nooro vs. other consumer NMES and EMS foot devices
The market for at-home foot stimulation devices has grown significantly. The Nooro device competes with products like the FootRenew, various EMS foot pad devices at lower price points, and multi-modal devices that combine heat, compression, and electrical stimulation. MBK has not conducted independent comparative testing. The Nooro distinguishes itself through its confirmed FDA 510(k) clearance, eight-mode NMES-specific programming, the peer-reviewed study citation, and the promotional price point with a ninety-day guarantee.
Nooro vs. lifestyle modifications
Regular walking activates the calf pump naturally. Foot elevation reduces swelling and supports venous return. These approaches complement rather than compete with a device. For people whose symptoms make walking difficult or painful, NMES provides a passive way to engage some of the same circulatory mechanisms that walking would otherwise activate - which is one reason the clinical research on NMES has focused on limited-mobility populations as a primary use case. Incorporating the device into a daily routine that also includes whatever level of activity is possible for your specific situation is likely more effective than relying on the device alone.
A note on multi-modal devices
The consumer foot device market has expanded to include products that combine NMES or EMS with heat therapy, air compression, or vibration. Multi-modal devices address multiple comfort dimensions simultaneously and may suit people who want more comprehensive lower-limb support beyond electrical stimulation alone. The Nooro device focuses on NMES as its primary mechanism without a built-in heat or compression component. Whether that focused approach or a multi-modal alternative is more appropriate depends on which aspects of your foot and leg discomfort matter most - a conversation worth having with your physician or physical therapist when choosing between device categories.
Spring 2026: Why Right Now Matters for People With Foot Pain
There is something specific about this moment in the seasonal calendar that matters for anyone evaluating this device.
The New Year resolution cycle for most health categories fades by February. Neuropathy and chronic pain categories are different. The motivation to do something about it does not disappear - it resurfaces every time contrast hits. Right now, entering spring 2026, that contrast is acute. Warmer weather is arriving. People want to walk outside, garden, travel, spend time with grandchildren at the park. And for people with significant neuropathic foot symptoms, every one of those activities involves a calculation about how much it is going to cost them.
That gap between what spring invites and what the feet allow is one of the sharpest emotional triggers in this product category. It is not just physical discomfort. It is the accumulating frustration of watching another season arrive with the same limitation still in place.
A ninety-day evaluation window beginning in mid-April runs through mid-July. That covers the entire prime spring and early summer activity window. If the device helps, you have the season to benefit from it. If it does not, you have the guarantee window to reach that determination before summer ends. The logic of trying it now rather than later is a practical one, not a manufactured sales argument.
Where Consumer Devices Fit in Neuropathy Management
Peripheral neuropathy affects an estimated twenty million people in the United States. Clinical management begins with identifying and addressing underlying causes: glycemic control for diabetic neuropathy, nutritional correction for B12 deficiency, and so on. Pharmacological symptom management, physical therapy, and lifestyle modification are the established clinical pillars.
Prescription medications used in neuropathic symptom management include anticonvulsants such as gabapentin and pregabalin, certain antidepressants including duloxetine, and topical agents including lidocaine and capsaicin formulations. Each works through pharmacological mechanisms, requires physician prescribing, and carries its own tolerability profile. For many people these medications provide meaningful symptom reduction. For others, side effects or incomplete efficacy motivate the search for complementary approaches.
Physical therapy, including supervised electrical stimulation with clinical-grade equipment, remains the gold standard for device-based neuropathy management. The clinical setting provides individualized parameter selection, professional monitoring, and adaptive protocol adjustment over time. The tradeoffs are cost, scheduling, and the fact that professional sessions are finite while neuropathic symptoms are often daily and ongoing.
Consumer electrical stimulation devices occupy a supplemental category alongside these pillars: daily wellness tools that may support circulation between professional appointments, used with physician awareness, as part of a broader approach. The research backing for NMES in lower-limb circulation contexts has a meaningful foundation in the rehabilitation literature. The consumer versions of this technology lack clinical oversight and individualized protocols - that is the meaningful tradeoff - but they do not lack a legitimate mechanism or, in the case of the Nooro device, a legitimate regulatory foundation.
The device most likely to provide value is one used by a person who has established appropriate medical management of their condition, understands the contributing factors in their case, and is adding daily NMES as a supplemental wellness tool. That is the context in which this guide recommends evaluating this category.
It is also worth noting that regular walking, when possible, activates the calf muscle pump naturally and is one of the most effective low-cost tools for lower-limb circulation. Foot elevation helps reduce swelling and supports venous return. These lifestyle approaches complement rather than compete with a device. For people whose symptoms make extended walking difficult or painful, NMES offers a passive way to engage some of the same circulatory mechanisms that walking would otherwise activate - which is part of why the clinical research on NMES has focused on immobile or limited-mobility patient populations as a primary use case.
Regarding the regulatory landscape: the consumer electrical stimulation device category has received ongoing attention from regulators regarding how devices are marketed relative to their cleared indications. Consumers should confirm any device's current regulatory standing and consult their healthcare provider before use.
This is not medical advice. Consult your physician for guidance appropriate to your specific situation.
Understanding the FDA 510(k) Clearance: What It Means for You
Because FDA clearance is one of the most searched questions for this product, and because the distinction between clearance and approval matters in ways that affect how you interpret the device's claims, this section walks through it clearly.
FDA 510(k) clearance is the regulatory pathway for medical devices that are substantially equivalent to a device that was already legally marketed before the Medical Device Amendments of 1976, or to a device that has itself received 510(k) clearance. The manufacturer submits data demonstrating that their device is substantially equivalent to this predicate device in intended use and technological characteristics.
What 510(k) clearance means in practical terms for the Nooro device: the FDA reviewed submission K192248 in 2019 and determined that the "Sumsens TENS and PMS" device, model SM9187, is substantially equivalent to its predicate in terms of safety and effectiveness for its cleared indication category. The device is cleared as a Class II medical device, meaning it is considered to have moderate risk and can be marketed with general controls and special controls.
What 510(k) clearance does not mean: the FDA did not independently test the specific marketing claims the Nooro brand makes about neuropathy relief, circulation improvement, or nerve recovery. The clearance applies to the safety profile of the device category, not to the specific outcome promises in the brand's advertising. This is an important distinction that competitors and critical reviews have noted, and this guide surfaces it directly because a reader who understands it is better positioned to evaluate the device honestly.
The practical upshot: the device's FDA clearance means it has passed a recognized regulatory pathway and is not being sold outside the regulatory system. It adds legitimacy to the brand's foundation. It does not transform marketing language into clinically proven outcomes. Both things can be true simultaneously, and both are relevant to your decision.
Who This Device May Be Right For: Self-Assessment Framework
This Device May Align Well With People Who:
Have physician awareness of their neuropathic condition and understand what is contributing to their symptoms. The ideal candidate has appropriate medical management in place and is exploring supplemental at-home tools - not using a consumer device as a substitute for diagnosis.
Are specifically dealing with circulation-related contributions to their neuropathic symptoms. The NMES mechanism is most directly relevant for presentations where vascular factors are recognized contributors.
Want a drug-free daily wellness tool to complement their existing management approach. The device operates on a different pathway from pharmacological interventions and does not create medication interactions at the device level.
Can commit to daily fifteen-minute sessions for at least sixty to ninety days. The research was conducted over eight weeks of consistent use. Consumer devices need comparable commitment to give a fair evaluation.
Are purchasing a one-time product and are comfortable with the return shipping cost if the device does not work for them. The ninety-day guarantee on one-time purchases is real and documented. Return shipping is the buyer's responsibility.
Are buying as a gift for someone with chronic foot discomfort. The spring timing, the practical design, and the guarantee make this a thoughtful option in a category with genuine scientific roots.
Other Options May Be Better Suited For People Who:
Have not been evaluated by a physician for their foot symptoms. Medical evaluation is the necessary first step.
Have a pacemaker, ICD, or other implanted electronic device. This is a firm contraindication for the entire electrical stimulation category. Do not use without explicit cardiologist clearance.
Are pregnant. Consult your physician before use.
Have active skin breakdown, ulceration, or infection on the feet. Do not apply to affected areas.
Are expecting dramatic reversal of established neuropathic damage. This is a wellness tool that supports circulation. It is not a cure for any form of neuropathy.
Are primarily interested in deep physical kneading. NMES delivers electrical stimulation, not pressure massage. The sensation is entirely different.
Questions to Ask Yourself First:
Have I discussed my symptoms with a physician and do I understand what is driving them? Am I buying a one-time product or a subscription, and do I understand the difference in guarantee terms? Can I commit to daily use for at least sixty days? Am I comfortable with return shipping costs if the device does not deliver? Have I noted the BBB rating and factored that into my expectations around customer service if an issue arises?
How to Get Started
According to the brand's official website, the device is sold exclusively through their direct channel and is not available on Amazon or eBay. Any marketplace listings should be approached with caution given the brand's own warning about impersonators.
One-time purchase is covered by the ninety-day money-back guarantee. Subscription options carry different terms - review carefully before enrolling.
Final Verdict: The Honest Bottom Line for Spring 2026
Here is the complete, balanced picture.
The case for trying the Nooro Whole Body Massager:
The device holds confirmed FDA 510(k) clearance as a Class II medical device. The NMES technology it uses has genuine peer-reviewed research support for the circulatory mechanism it operates on. The specific PubMed citation the brand provides is a real study published in the scientific literature. The device's feature set is solid at the current promotional price. The ninety-day money-back guarantee on one-time purchases creates a bounded evaluation window. The brand publishes verifiable corporate information and contact details.
Considerations to weigh before ordering:
The brand's parent company carries an F rating with the Better Business Bureau, with documented complaints about refund processing and product effectiveness. The marketing language includes framing like "treat the real root cause" and "nerves can finally repair and heal" - these go beyond what the cited research specifically proves for this device as a finished product. The research validates the technology category, not guaranteed individual outcomes. Return shipping is the buyer's cost. Subscription products carry more restrictive guarantee terms. And the practical reality of neuropathic conditions is that no consumer device addresses every presentation.
Who should try it: Adults with neuropathic foot symptoms who have physician awareness of their condition, who understand they are evaluating a supplemental wellness tool with FDA clearance and a legitimate research foundation, who can commit to daily use, and who have factored in the BBB rating and return shipping when assessing their risk. For that person, the price point and guarantee structure make a ninety-day evaluation reasonable.
See the current Nooro Whole Body Massager offer on the official website
Frequently Asked Questions
Q: Is the Nooro Whole Body Massager FDA approved or cleared?
According to publicly available FDA records and the brand's own product documentation, the device holds 510(k) clearance as a Class II medical device under submission K192248, issued in 2019. The underlying device is identified as "Sumsens TENS and PMS," model SM9187. FDA 510(k) clearance means the agency determined the device is substantially equivalent to a legally marketed predicate device in its category - it is not the same as FDA approval for a specific therapeutic indication, and it does not validate the brand's specific marketing claims about neuropathy outcomes. Consumers can verify the clearance independently through the FDA's 510(k) database.
Q: Is the Nooro Whole Body Massager the same as a TENS unit?
No. TENS targets sensory nerves to modulate pain signals. NMES targets motor nerves to induce muscle contraction. In the foot and calf context, NMES activates the musculo-venous pump through calf contraction - a circulatory mechanism that standard TENS does not replicate at the same level. The distinction is supported in the rehabilitation literature, not just in the brand's marketing.
Q: How soon might I notice a change?
According to the brand's FAQ, some sensation is perceptible during the first use. For changes related to ongoing use, the brand suggests some users begin noticing differences after approximately two weeks of consistent daily use. These are the brand's usage guidance timelines, not guaranteed outcomes. Individual experiences vary considerably based on symptom type, severity, and consistency of use.
Q: Can I use this device if I have diabetes?
Diabetes involves specific considerations for electrical stimulation devices, including skin sensitivity, sensation changes, and cardiovascular factors. Always consult your physician before using any NMES or TENS device if you have diabetes.
Q: Can I use this if I have a pacemaker?
No. Do not use any electrical stimulation device if you have a pacemaker, ICD, or any other implanted electronic medical device without explicit physician clearance. This is a firm contraindication for the entire category.
Q: Does the 90-day guarantee cover subscriptions?
No. The ninety-day money-back guarantee applies only to one-time physical product purchases through the official website. Subscription renewals are explicitly excluded per the brand's official refund policy. If the guarantee matters to your decision, purchase the one-time product option.
Q: What is the BBB rating situation?
XF Agencija US Inc., the company behind Nooro, carries an F rating with the Better Business Bureau, with publicly visible complaints centered on product effectiveness and refund processing. This does not invalidate the device's FDA clearance or the underlying technology. It does mean that if you need to use the return process, patience and documentation of your communication will serve you better than assuming it will be frictionless.
Q: Why is it not on Amazon?
According to the brand's product page, the device is sold exclusively through their official website. The brand specifically warns that other companies misrepresent themselves as Nooro on third-party platforms. Any Amazon or eBay listings for "Nooro" should be treated with caution.
Q: How long do the pads last and what do replacements cost?
According to the brand's listing, each set of two pads is rated for approximately one month of daily use. Replacement sets cost approximately ten dollars per the brand's listing. Verify current pricing on the official website.
Q: Is it safe for swollen ankles and feet?
The brand describes swollen feet and ankles as one of the conditions the device is designed to address, and the circulatory mechanism is directly relevant to edema related to poor circulation or prolonged immobility. For significant edema associated with cardiac, renal, or lymphatic conditions, consult your physician first to ensure the underlying cause is understood and managed.
Q: Can chemo patients use this?
Chemotherapy-induced peripheral neuropathy is a recognized condition that the NMES mechanism may be relevant to discuss with an oncology care team. Whether this device is appropriate for someone actively in treatment or post-treatment is a decision that belongs with the treating oncologist.
Q: What is the return process?
Contact support@nooro-us.com or the Help Center within ninety days of your order date. Follow the return instructions from the support team. Return shipping is your cost - the brand does not provide prepaid labels. Refunds process within seven to ten business days after receipt and inspection. Given the F BBB rating, document your communications throughout the process.
Q: I have been using a TENS unit for years. Is this worth trying?
If TENS has been providing temporary relief but limited lasting effect, and you are specifically interested in the circulatory mechanism, NMES operates through motor nerve activation and calf contraction in a way that TENS does not replicate. That mechanistic difference is a scientifically grounded reason to evaluate NMES separately. Discuss any changes to your management approach with your physician.
Q: Is the Nooro Whole Body Massager worth it at the current price?
At the current promotional price, the device sits at roughly the cost of one podiatry copay for many people with insurance, and far less than one out-of-pocket appointment. For a device with confirmed FDA clearance, peer-reviewed technology backing, and a ninety-day guarantee on one-time purchases, the price point makes a trial financially reasonable for the right person. The right person has physician awareness of their condition, realistic expectations about what a supplemental wellness device can deliver, the ability to commit to daily use, and has factored in the BBB rating and return shipping as part of the risk calculation.
Q: Can I use this for plantar fasciitis?
The brand does not specifically market the device for plantar fasciitis. Plantar fasciitis involves inflammation of the plantar fascia connective tissue along the bottom of the foot and is a distinct condition from peripheral neuropathy or circulatory insufficiency. The device may provide some comfort during use for general foot discomfort, but plantar fasciitis has specific management approaches - stretching protocols, supportive footwear, physical therapy, and in some cases orthotics or cortisone injections - that should be discussed with a podiatrist or sports medicine physician. Using an NMES device is not a substitute for that evaluation.
Q: Is the Nooro Whole Body Massager FSA or HSA eligible?
FSA and HSA eligibility for consumer NMES devices varies by plan and administrator and is not universally guaranteed. The device holds FDA clearance as a Class II medical device, which may support eligibility claims, but individual plan rules determine what qualifies. The Nooro brand's materials do not make explicit FSA or HSA eligibility claims in the product documentation reviewed for this article. Verify directly with your plan administrator before purchasing with the assumption of reimbursement.
Q: How much does it cost to maintain the device over a year?
Based on the brand's pricing as of April 2026, the device costs approximately $49.95 upfront at the current promotional price. Replacement adhesive pads cost approximately $10 per set per month for daily users, per the brand's listing. Over twelve months of daily use, that works out to approximately $170 total - roughly $49.95 for the device plus approximately $120 in replacement pads. The brand's estimate of roughly forty-seven cents per session assumes daily fifteen-minute use at that pad replacement rate. All pricing is subject to change; verify current replacement pad pricing on the official website.
Q: Can I use this on my calves and ankles as well as my feet?
Yes. According to the brand's product page and usage instructions, the device is designed for application on feet, ankles, and calves together, which the brand recommends specifically to address the full lower-extremity circulation chain. The brand describes the device as a whole body massager with a broader application range, though the primary marketing focus is the foot and lower leg area relevant to neuropathic symptoms and circulation support.
Q: Does it work for numbness, or just burning and tingling?
The brand's positioning addresses burning, tingling, and numbness as a cluster of neuropathic symptoms. The circulatory mechanism the device operates on - NMES-induced calf contraction activating the musculo-venous pump - does not target specific symptom types differently. It supports lower-limb blood flow generally, which the brand premises may contribute to perceived comfort across different neuropathic sensations. Whether any individual experiences changes in numbness specifically, rather than burning or tingling, is not something this guide can predict, and the brand does not separate outcomes by symptom type in its materials. If numbness is your primary concern, discuss with your physician whether a circulatory support approach is relevant to your specific presentation.
Q: Is this safe to use every day long-term?
The brand recommends daily use as the standard protocol, with fifteen-minute sessions as the target. NMES devices used at consumer intensity levels are generally considered well-tolerated for regular use when standard contraindications are respected. The primary practical considerations for long-term daily use are pad adhesion quality over time - which is why replacement pads are sold separately - and skin integrity at application sites. If you notice any skin irritation, redness, or breakdown at the pad placement areas, discontinue use at that site and consult your physician. The brand does not publish explicit guidance on the maximum recommended duration of continuous daily use in months or years; for long-term use questions, consult your physician.
Q: Who makes this device?
The Nooro Whole Body Massager is sold by XF Agencija US Inc., a Delaware-registered company with EIN 93-1616450. The underlying device is manufactured by Hang Zhou Anqing Shen Zhen Electronics Limited under the product name "Sumsens TENS and PMS," model SM9187, and is distributed under the Nooro brand name in the US market.
Visit the official Nooro website to check current pricing, availability, and product details
Customer Support
According to the company's official website as of April 2026:
Company: Nooro
Phone: +1 212-444-3144
Email: support@nooro-us.com
Company address: XF Agencija US Inc., 1111B S Governors Ave Number 80212, Dover, DE 19904
Contact information is attributed to the brand's official website as of April 2026. Verify current availability directly with the brand.
Related: Nooro Foot Massager Review
Disclaimers
Editorial Disclaimer: This article is for informational purposes only and does not constitute medical advice, diagnosis, or treatment recommendation. The information presented reflects publicly available details from the Nooro brand's official website and legal pages, publicly available FDA records, and general scientific and industry context. Always verify current terms, pricing, and product details directly with Nooro before purchasing.
Medical Disclaimer: This article is educational and does not constitute medical advice. The Nooro Whole Body Massager is a consumer device and is not a substitute for professional medical evaluation or treatment. If you are experiencing neuropathic symptoms, foot pain, circulation concerns, or any health condition addressed in this article, consult a licensed healthcare professional before using this or any other device. Do not change, adjust, or discontinue any prescribed medical treatments without your physician's guidance and approval. This device is not a replacement for prescribed medical treatment for any health condition.
Results May Vary: Individual results will vary based on factors including the nature and severity of symptoms, consistency of device use, underlying health conditions, current medications, age, baseline circulation health, and other individual variables. The research cited in this article relates to NMES technology in general and does not constitute proof of specific outcomes for the Nooro Whole Body Massager as a finished consumer product. Some users report experiencing meaningful changes; others do not. Results are not guaranteed.
Device Safety Notice: Do not use this or any electrical stimulation device if you have a pacemaker, implanted cardioverter-defibrillator, or other implanted electronic medical device without explicit physician clearance. Do not use on broken, irritated, or infected skin. Consult your physician before use if you are pregnant, have heart disease, diabetes, or any serious medical condition.
FTC Affiliate Disclosure: This article contains affiliate links. If you purchase through these links, a commission may be earned at no additional cost to you. This compensation does not influence the accuracy, neutrality, or integrity of the information presented. All descriptions are based on publicly available information from the brand's official website, public FDA records, and general scientific context.
Pricing Disclaimer: All pricing information was accurate based on publicly available information at the time of publication (April 2026) and is subject to change without notice. Always verify current pricing, bundle options, and promotional terms on the official Nooro website before completing your purchase.
Guarantee Disclaimer: The ninety-day money-back guarantee applies to one-time physical product purchases made directly through the official Nooro website, per the brand's published refund policy as of April 2026. Subscription products are explicitly excluded. Return shipping costs are the responsibility of the customer. Always review current refund and return terms on the official Nooro website before purchasing.
Publisher Responsibility Disclaimer: The publisher of this article has made every effort to ensure accuracy at the time of publication based on publicly available information. We do not accept responsibility for errors, omissions, or outcomes resulting from the use of the information provided. Readers are encouraged to verify all details directly with Nooro and their healthcare provider before making decisions.
SOURCE: Nooro
Source: Nooro
