New Research Finds that Sustained Acoustic Medicine (sam®) Treatment Significantly Reduced Pain of the Neck and Shoulder
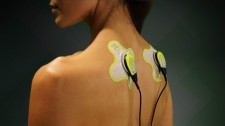
TRUMBULL, Conn., June 16, 2020 (Newswire.com) - ZetrOZ Systems, maker of wearable ultrasound device sam®, is encouraged by a double-blind RCT study published recently in the Journal of Pain Research by the Orthopaedic Foundation, Stamford, Connecticut, found that low-intensity continuous ultrasound treatment significantly reduced upper trapezius myofascial pain of the neck and shoulder. Results indicated that applying daily continuous ultrasound treatment of 18,720 joules delivered by a wearable sustained acoustic medicine (sam®) device caused a clinically meaningful improvement in the global rate of change (GROC) scores for patients.
Low-intensity continuous ultrasound is an emerging high-dosimetry low-intensity continuous ultrasound-based therapy that facilitates accelerated tissue healing and treats myofascial pain. In this instance, low-intensity continuous ultrasound treatment was evaluated for chronic upper neck and shoulder pain in a randomized, multi-site, double-blind, placebo-controlled study.
The level 1 study followed the nationally recognized Consolidated Standards of Reporting Trials (CONSORT) guidelines that were used to recruit and conduct the clinical efficacy trial. Thirty-three patients with upper trapezius myofascial pain were randomized for treatment with active (n=25) or placebo (n=8) devices. Investigators and subjects were blinded to treatment groups. Patients self-reported their pain level daily, rating it from zero to 10 on the numeric rating scale (NRS).
If pain rating was more significant than or equal to three, the sam® device (3MHz,0.132W/cm2,1.3W, four hours) was self-applied for total energy dosimetry of 18,720 joules per treatment. During the four-week study, the daily pain rating was recorded. If sam®-treatment was delivered, pain before, during and after treatment was recorded along with the global rate of change (GROC). Statistical analysis was used to assess changes from the baseline and differences between treatment groups.
Patients treated with active sam® treatment observed a significant mean pain reduction of 2.61-points for active (p<0.001), compared to 1.58-points decrease from baseline for placebo (p=0.087) - resulting in a 1.03-point significant decrease in the active group over placebo (p=0.003). The overall GROC was significantly higher in the active group at 2.84 points compared to the placebo group at 0.46 points (p<0.001).
"We are greatly encouraged by the results of this double-blind RCT study. The clinical outcome confirms the value to patients, providers and payers who use sam® to relieve pain and reduce medication, including upper trapezius myofascial pain of the neck and shoulder. sam® does not just reduce pain, but the device also has the ability to treat arthritis and soft tissue injuries," said Dr. George Lewis, chief executive officer of ZetrOZ Systems.
Reference to Study
Petterson S,1Plancher K,1-4Klyve D,5 Draper D,6 Ortiz R.7 Low-Intensity Continuous Ultrasound for the Symptomatic Treatment of Upper Shoulder and Neck Pain: A Randomized, Double-Blind Placebo-Controlled Clinical Trial. J Pain Res. 2020;13:1277-1287
https://doi.org/10.2147/JPR.S247463
1Orthopaedic Foundation, Stamford, CT, USA; 2Department of Orthopaedics, Albert Einstein College of Medicine, New York, NY, USA; 3Department of Orthopaedics, Weill Cornell Medical College, New York, NY, USA; 4Plancher Orthopaedics & Sports Medicine, New York, NY 10128, USA; 5Department of Mathematics, Central Washington University, Ellensburg, WI, USA; 6Department of Exercise Sciences, Brigham Young University, Provo, UT, USA; 7Medical Pain Consultants, Dryden, NY, USA
About ZetrOZ Systems
ZetrOZ Systems is an FDA cGMP and ISO 13585 medical technology company headquartered in the southern coastal region of Connecticut. The organization also has manufacturing facilities across the United States. ZetrOZ Systems produced UltrOZ®, sam®Sport and sam®Pro 2.0 to provide safe and effective treatment options for prevalent conditions such as arthritis. Learn more at zetroz.com and samrecover.com.
Source: ZetrOZ Systems
