FDA Created New Category for ZetrOZ's Long-Duration Pain Management Ultrasound Device
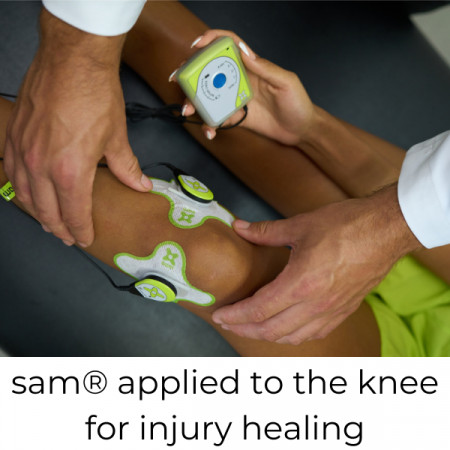
TRUMBULL, Conn., September 29, 2021 (Newswire.com) - ZetrOZ System's Sustained Acoustic Medicine (sam®) device, the only FDA-cleared device of its kind, is part of a new category established by the U.S. Government for the novel device's wearable, long-duration ultrasound therapy for home use.
A Class II medical device initially cleared by the FDA in 2013, sam® was later provided with expanded indications in 2020. It is the only home-use, long-duration ultrasound treatment for reducing pain and improving circulation to heal soft tissue injuries.
"It is gratifying that after starting to develop the medical innovation nearly 15 years ago, we have successfully worked with the FDA to elucidate our sam® technology and build a strong portfolio of clinical evidence to support the safety and efficacy of our device," says George K. Lewis, Ph.D., founder, president, and CEO of ZetrOZ Systems. "By teaming up with a superb group of researchers, we developed a treatment algorithm that was able to be applied to people and stimulate soft tissue repair."
From basic science studies in the lab to small and then large animal studies, the ZetrOZ team ultimately commenced clinical trials to determine the safety, efficacy, and clinical utility for human patients. The result was sam®, a noninvasive mechanobiologic product for in-home use that involves no drugs and reduces the need for pain medication or surgery.
Now clinically validated in over 30 scientific studies with more than 300,000 patients successfully treated, the sam® device addresses the regenerative medicine market, improving postoperative recovery and joint function.
sam® generates deep heat, increases local circulation, and activates cellular pathways that help the body heal after an injury, which ultimately leads to a faster return to work, sports or other activities. The sam® treatment is typically applied once per day for six to eight weeks as prescribed by a healthcare provider.
With sam®, the future is here … a clinically validated, in-home ultrasound therapy that speeds healing for those suffering with painful conditions such as arthritis, inflammation, tendon injury and more.
For additional information, please visit samrecover.com or zetrozsystems.com.
About ZetrOZ Systems
ZetrOZ Systems is an FDA cGMP and ISO 13585 medical technology company headquartered in the southern coastal region of Connecticut. The organization also has manufacturing facilities across the United States. ZetrOZ Systems produced UltrOZ®, sam®Sport and sam®Pro 2.0 to provide safe and effective treatment options for prevalent conditions such as arthritis. Learn more at zetroz.com and samrecover.com.
Media Contact:
Bianca D'Angelo
(203) 577-7588 (Direct)
bianca@newswire.com
www.Newswire.com
Source: ZetrOZ Systems
