eDSMB® Expands Ethical's Portfolio of Secure, Compliant Software for Clinical Committee Management
BASEL, Switzerland, September 20, 2022 (Newswire.com) - Ethical GmbH, a Swiss eClinical company providing cost-effective, compliant software and customized support to simplify the daily work of clinical teams, is releasing a new software for the management of DSMBs, DMCs and Steering Committees.
The use of clinical committees such as DSMBs, DMCs and Steering Committees to increase the quality of clinical trials has become common practice. Managing a panel of experts, however, requires performing time-consuming, critical activities such as planning meetings, presenting clean data, preventing accidental unblinding, signing meeting reports, filing decisions, etc. A secure, efficient process supported by an easy-to-use tool is essential to guarantee the trial quality and overcome time constraints.
One single hub
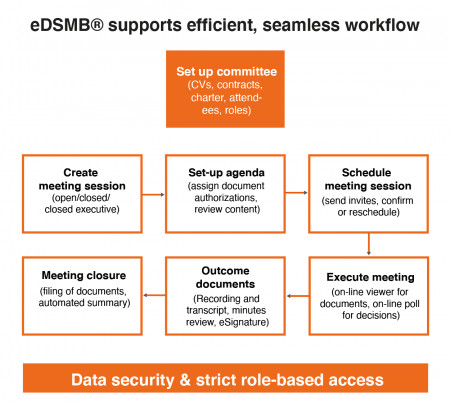
eDSMB® is a cloud-based platform supporting all operations related to a DSMB, DMC or Steering Committee. Using a single platform and a single repository, clinical teams collect committee members' CVs, schedule meetings, collaborate online, send invites, manage authorizations, conduct meetings, collect e-signatures, document decisions, export for TMF, etc.
Security, efficiency and compliance
eDSMB® provides a model for best-practice workflow through document and meeting templates, role-based authorizations, secured access, assistance to configure, plan and execute meetings and other features. Audit trails of all operations performed on the system are available at any time for compliance inspections. (www.edsmb.com)
Mimmo Garibbo, Director of Ethical GmbH stated: "With eDSMB®, Ethical continues to expand its portfolio of secure and compliant software solutions for the management of clinical committees. With eAdjudication® for clinical adjudication, eDeviation® for protocol deviations, and now eDSMB® for the management of DSMBs, our clients benefit from a full range of easy-to-use software platforms for managing their clinical committees in a more efficient way."
Interested in seeing a Demo? Make a request here.
Download the Complete DSMBs-DMCs Handbook here (20 pages).
Source: Ethical GmbH
