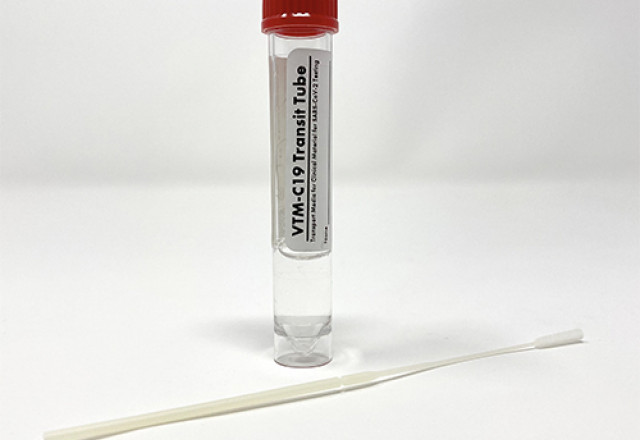
Biomed Diagnostics Viral Transport Media for COVID-19 Now Available

MEDFORD, Ore., March 8, 2021 (Newswire.com) - Biomed Diagnostics, a medical device innovator and manufacturer, is pleased to announce the commercial availability of its viral transport medium (VTM) to support testing in the US and Canada for the virus causing COVID-19 disease.
Biomed has significantly increased production volume of two transport media devices: VTM-C19™ and Saline Solution (.85%)™. Both formulations have been validated for the safe transport and preservation of specimens from patients being tested for SARS-CoV-2 (COVID-19), influenza A, and RSV.
Both VTM-C19 and Saline Solution (.85%) are manufactured by Biomed in their ISO 13485:2016 compliant, FDA-registered production facility, in Oregon, USA. Both are available through the US and Canada, listed with the FDA and Health Canada.
VTM-C19 is listed with the FDA and compliant with the FDA EUA policy for use in specimen collection and transport, and compliant with FDA-cleared testing as described in the FDA guidance for molecular Influenza and RSV and COVID-19. The media is formulated as recommended by the CDC for the transportation of specimens from patients suspected of a COVID-19 infection.
VTM-C19 Transit Tube (3 ml) can be stored at room temperature (18-25°C) for up to 12 months; Biomed guarantees product quality with a QC audit of each lot through its expiration. The tube is intended for inoculation of specimens from a nasopharyngeal (NP) or oropharyngeal (OP) synthetic fiber swab.
Saline Solution (0.85%) 3ml is registered and listed with the FDA, and Biomed R&D labs have validated the solution to ensure a non-replicating competent status of SARS-CoV-2 for qRT-PCR diagnostic testing. SARS-CoV-2 virus stability has been validated in the Saline Solution for Room Temperature storage and for incubation for up to 72 hours post inoculation. The tube is intended for inoculation of specimens with nasopharyngeal (NP) or oropharyngeal (OP) synthetic fiber swab.
According to the Rockefeller Foundation, which is providing over $50 million toward COVID testing and tracing, upwards of 30 million tests are needed per week in the U.S. alone.
For more information, please visit Biomed Diagnostics, or call 800.964.6466.
About Biomed Diagnostics
Biomed Diagnostics, Inc. is an FDA-registered production facility, headquartered in Oregon, USA, and designs, engineers and manufactures prepared culture media devices for the collection, propagation, and detection of pathogenic organisms. Biomed distributes products to over 60 countries, and partners with numerous humanitarian organizations to diagnose and prevent disease and protect both animal and human health.
Biomed Diagnostics operates in compliance with FDA Quality System (QS) Regulation/Medical Device Good Manufacturing Practices (21 CFR, Part 820) in the manufacture of all products.
Biomed Diagnostics COVID testing supplies
Saline Solution (.85%) Product Data Sheet
Source: Biomed Diagnostics